
Rather, he wrote that, '. elements of like properties or their compounds of like properties, form groups bounded by simple lines. In his article Hinrichs included a periodic table, organized by atomic weight, but this did not show a metal-nonmetal dividing line. Deming himself noted that the line could not be drawn very accurately. Several other sources note confusion or ambiguity as to the location of the dividing line suggest its apparent arbitrariness provides grounds for refuting its validity and comment as to its misleading, contentious or approximate nature. impossible to draw a strict line of demarcation between metals and nonmetals, there being many intermediate substances'. One line separates metals and metalloids the other metalloids and nonmetals. Double line variant Ī dividing line between metals and nonmetals is sometimes replaced by two dividing lines. It was also distributed for many years by the Sargent-Welch Scientific Company. By the 1930s Deming's table was appearing in handbooks and encyclopaedias of chemistry. Merck and Company prepared a handout form of Deming's 18-column table, in 1928, which was widely circulated in American schools. Each one had a regular stepped line separating metals from nonmetals. Deming, an American chemist, published short ( Mendeleev style) and medium ( 18-column) form periodic tables. In 1906, Alexander Smith published a periodic table with a zigzag line separating the nonmetals from the rest of elements, in his highly influential textbook Introduction to General Inorganic Chemistry. In 1891, Walker published a periodic 'tabulation' with a diagonal straight line drawn between the metals and the nonmetals. References to a dividing line between metals and nonmetals appear in the literature as far back as at least 1869. The former generally combine with electropositive metals to make intermetallic compounds whereas the latter usually form salt-like compounds. 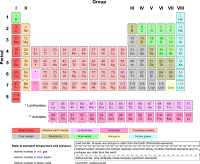
It differentiates group 13 elements from those in and to the right of group 14. This particular line was named by Laves in 1941. While it has also been called the Zintl border or the Zintl line these terms instead refer to a vertical line sometimes drawn between groups 13 and 14. This line has been called the amphoteric line, the metal-nonmetal line, the metalloid line, the semimetal line, or the staircase. When presented as a regular stair-step, elements with the highest critical temperature for their groups (Li, Be, Al, Ge, Sb, Po) lie just below the line. Elements to the lower left of the line generally display increasing metallic behaviour elements to the upper right display increasing nonmetallic behaviour. The dividing line between metals and nonmetals can be found, in varying configurations, on some representations of the periodic table of the elements (see mini-example, right).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
